A Direct Reduction of Aliphatic Aldehyde, Acyl Chloride, Ester, and Carboxylic Functions into a Methyl Group
Vladimir Gevorgyan*, Michael Rubin, Jian-Xiu Liu and Yoshinori Yamamoto
*Department of Chemistry, University of Illinois at Chicago, 845 W. Taylor
Street, Chicago, Illinois 60607-7061, Email: vlad uic.edu
uic.edu
V. Gevorgyan, M. Rubin, J.-X. Liu, Y. Yamamoto, J. Org. Chem, 2001, 66, 1672-1675.
DOI: 10.1021/jo001258a


Abstract
Aliphatic carboxyl derivatives (acids, acyl chlorides, esters) and aldehydes were efficiently reduced to the methyl group by HSiEt3 in the presence of catalytic amounts of B(C6F5)3. Aromatic carboxylic acids, as well as other carbonyl functional equivalents, underwent smooth partial reduction to the corresponding TES-protected benzylic alcohols.
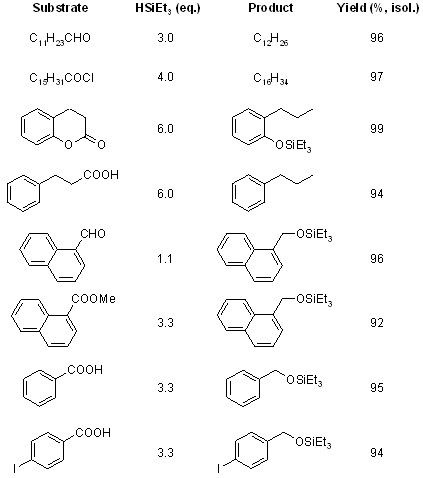 see article for more examples
see article for more examples
Key Words
Deoxygenations, Alcohol Synthesis, Silyl Ethers, Triethylsilane
ID: J42-Y2001-570
