Categories: C-C Bond Formation > Arenes >
Synthesis of aryl ketones
| Related: |
|
|
|
|
|
Name Reactions
Recent Literature

Stable and non-hygroscopic aluminum
dodecatungstophosphate (AlPW12O40), which is prepared
easily from cheap and commercially available compounds, is an
effective catalyst for Friedel-Crafts acylation reactions using carboxylic
acids, acetic anhydride and benzoyl chloride in the absence of solvent under
mild reaction conditions.
H. Firouzabadi, N. Iranpoor, F. Nowrouzi, Tetrahedron, 2004,
60, 10843-10850.

A synergistic merger of photoredox, nickel, and hydrogen atom transfer catalysis
enables direct aldehyde C-H functionalization. This mild, operationally simple
protocol provides a broad range of ketones in excellent yield from commercially
available aldehydes, along with aryl or alkyl bromides.
X. Zhang, D. W. C. MacMillan, J. Am. Chem. Soc., 2017,
139, 11353-11356.

The cooperative catalysis of nickel and tetrabutylammonium decatungstate as a
hydrogen-atom-transfer photocatalyst enables a cross-coupling of aryl halides
and α-bromo acetates with an array of (hetero)aromatic and aliphatic aldehydes
to provide a variety of ketones. This method tolerates a broad range of functional groups.
P. Fan, C. Zhang, L. Zhang, C. Wang,
Org. Lett., 2020, 22, 3875-3878.

The palladium acetate-catalyzed coupling reaction of aryl boronic acids with
carboxylic anhydrides or acyl chlorides was carried out smoothly in water in
the presence of PEG or [bmim][PF6] to give high yields of
ketones. The Pd(OAc)2-H2O-[bmim][PF6]
catalytic system can be recovered and reused.
B. Xin, Yuhong Zhang, K. Cheng, J. Org. Chem., 2006,
71, 5725-5731.

A mechanochemical solvent-free acyl Suzuki-Miyaura cross-coupling enables a
highly chemoselective synthesis of ketones from widely available acyl chlorides
and boronic acids. This chemoselective acylation reaction is conducted in the
solid state, in the absence of potentially harmful solvents, and for a short
reaction time.
J. Zhang, P. Zhang, Y. Ma, M. Szostak, Org. Lett.,
2022, 24, 2338-2343.
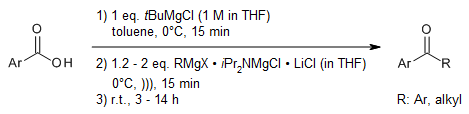
A magnesium amide additive activates and controls the addition of mild Grignard
reagents to carboxylate anions and therefore avoids the use of organolithium
reagents or activated acyl sources that need to be independently prepared. This
strategy enables the modular synthesis of ketones from CO2 and the
preparation of isotopically labeled pharmaceutical building blocks in a single
operation.
K. Colas, A. C. V. D. dos Santos, A. Mendoza,
Org. Lett., 2019, 21, 7851-7856.

[DCPP]n aggregates can act as a versatile visible-light
photocatalyst in decarboxylative C-C cross-coupling, amidation, and
esterification reactions. In these reactions, [DCPP]n aggregates were formed in
situ through physical π-π stacking of DCPP monomers in organic solvents.
M. He, X. Yu, Y. Wang, F. Li, M. Bao, J. Org. Chem., 2021, 86,
5016-5025.

The inexpensive 2-chloro-thioxanthen-9-one as photoredox catalyst mediates the
formation of acyl radicals from α-keto acids under visible light illumination.
These reactive species added to olefins or coupled with aryl halides via a
bipyridyl-stabilized Ni(II) catalyst to provide a diverse range of ketones. This
reliable, atom-economical, and eco-friendly protocol tolerates a wide range of
functional groups.
D.-L. Zhu, Q. Wu, D. J. Young, H. Wang, Z.-G. Ren, H.-X. Li,
Org. Lett., 2020, 22, 6832-6837.

Friedel-Crafts reactions of oxalyl chloride with two arenes provide symmetrical
and unsymmetrical diaryl ketones in good yields.
D. F. Taber, M. R. Sethuraman, J. Org. Chem., 2000,
65, 254-255.

Polysilane/alumina-supported palladium nanoparticles catalyze carbonylative
Suzuki-Miyaura coupling reactions under atmospheric pressure of CO gas to provide diaryl ketones in excellent yields. The ligand-free
immobilized palladium nanoparticles could be recovered by simple filtration and
reused.
T. Yasukawa, Z. Zhu, Y. Yamashita, S. Kobayashi, Synlett, 2021,
32,
502-504.

A palladium-catalyzed cross-coupling of the lithium anion of aryl tert-butyldimethylsilyl-protected
cyanohydrins with aryl bromides followed by in situ deprotection with fluoride
ion provides a convenient and versatile approach to biaryl ketones. This
protocol represents an example of a palladium-catalyzed arylation of a
cyanohydrin, which functions as an acyl anion equivalent.
J. Majhi, B. Zhou, Y. Zhung, M.-J. Tom, H. Dai, P. A. Evans, Synthesis, 2023,
55, 1752-1763.

Evaluation of N-acyl-imides, N-acyl-lactams, and N-acyl-oxazolidinones
bearing five- and six-membered rings using Pd(II)-NHC and Pd-phosphine systems
has
revealed the relative reactivity order of N-activating groups in
Suzuki-Miyaura cross-coupling. N-Acyl-δ-valerolactams are highly
effective precursors for cross-coupling via acyl-metal intermediates.
Md. M. Rahman, D. J. Pyle, E. Bisz, B. Dziuk, K. Ejsmont, R. Lalancette, Q.
Wang, H. Chen, R. Szostak, M. Szostak, J. Org. Chem., 2021, 86,
10455-10466.

A combination of site-selective N,N-di-Boc-activation of the amide
nitrogen with practical air- and moisture-stable, well-defined, and highly
reactive [Pd(NHC)(cin)Cl] enables a general, highly selective method for the
room temperature Suzuki-Miyaura cross-coupling to provide biaryl ketones in high
yields.
P. Lei, G. Meng, Y. Ling, J. An, S. P. Nolan, M. Szostak, Org. Lett.,
2017, 19, 6510-6513.

The commercially available [(IPr)PdCl2(aniline)] complex is a well-defined, air- and
moisture-stable catalyst for highly efficient Suzuki-Miyaura cross-coupling of
amides, esters, aryl chlorides, and Buchwald-Hartwig amination. The availability of various
aniline scaffolds has a significant potential in fine-tuning of challenging cross-couplings by Pd-NHCs.
Q. Xia, S. Shi, P. Gao, R. Lalancette, R. Szostak, M. Szostak, J. Org. Chem., 2021, 86,
15650-15657.

A highly chemoselective Suzuki-Miyaura cross-coupling of
trifluoromethanesulfonamides (triflamides) accomplishes a direct synthesis of
ketones under mild conditions as a powerful alternative to the Weinreb ketone
synthesis.
S. Shi, R. Lalancette, R. Szostak, M. Szostak, Org. Lett., 2019, 21,
1253-1257.

The reaction of different aryl, alkyl, and alkynyl Grignard reagents with
N-Boc amides provides a wide range of aryl ketones via chemoselective C(O)-N
bond cleavage under catalyst-free conditions. α-Ketoamide was successfully
converted to aryl diketones, while α,β-unsaturated amide underwent 1,4-addition
followed by C(O)-N bond cleavage to provide diaryl propiophenones.
P. Sureshbabu, S. Azeez, N. Muniyappan, S. Sabiah, J. Kandasamy, J. Org. Chem., 2019,
84, 11823-11838.

The combination of electrocatalysis and nickel catalysis achieves a
decarboxylative cross-coupling of four types of α-oxocarboxylic acids and their
derivatives with aryl trimethylammonium salts under mild conditions. This
strategy enables a practical synthesis of aryl ketones, amides, esters, and
aldehydes.
X. Kong, Y. Chen, X. Chen, Z.-X. Lu, W. Wang, S.-F. Ni, Z.-Y. Cao, Org. Lett.,
2022, 24, 2137-2142.

N-acylazetidines are bench-stable, readily available amide acylating
reagents, in which the reactivity is controlled by amide pyramidalization and
strain of the four-membered ring. A general and highly chemoselective synthesis
of ketones by the addition of organometallics to N-acylazetidines via
stable tetrahedral intermediates offers wide substrate scope and exquisite
selectivity for the ketone products.
C. Liu, M. Achtenhagen, M. Szostak, Org. Lett.,
2016, 18, 2375-2378.

Pd(OAc)2/PCy3 catalyzes the Hiyama cross-coupling
between N-acylglutarimides and arylsiloxanes in the presence of Et3N·3HF
and LiOAc to provide the corresponding arylketones in good yields. The reaction
had a broad substrate scope and showed good functional group tolerance.
M. A. Idris, S. Lee, Org. Lett., 2020, 22, 9184-9189.

A palladium-catalyzed Suzuki-Miyaura cross-coupling of
unconventional amide electrophiles enables a synthesis of sterically hindered
ketones via a selective N-C(O) activation. Mechanistic
studies demonstrate that steric bulk on the amide has a major impact, which is
opposite to the traditional Suzuki-Miyaura cross-coupling of sterically hindered
aryl halides.
C. Liu, R. Lalancette, R. Szostak, M. Szostak,
Org. Lett., 2019, 21, 7976-7981.

A simple method for the preparation of aryl methyl ketones involves the
Pd-catalyzed coupling of commercially available acetyltrimethylsilane with aryl bromides to afford the
corresponding acetylated arenes in very good yields. The methodology is tolerant
of heterocycles and provides a new method for arene functionalization.
S. D. Ramgren, N. K. Garg, Org. Lett., 2014,
16, 824-827.

A mild and general method for the palladium-catalyzed, copper-mediated coupling
of thiol esters and boronic acids under "baseless" conditions has been
developed. The general availability of both reaction partners and the nonbasic
reaction condition suggest new possibilities for the synthesis of highly
functionalized and base-sensitive compounds under these cross-coupling
conditions.
L. S. Liebeskind, J. Sorgl, J. Am. Chem. Soc., 2000,
122, 11260-11261.

A direct acylation of aryl bromides with aldehydes involves palladium-amine
cooperative catalysis, affording synthetically important alkyl aryl ketones in
good yields in a straightforward manner, and broadening the scope of
metal-catalyzed coupling reactions.
J. Ruan, O. Saidi, J. A. Iggo, J. Xiao, J. Am. Chem. Soc., 2008,
130, 10510-10511.

FeCl2 catalyzes a smooth and convenient acylation of functionalized
arylzinc halides and benzylic zinc chlorides with various acid chlorides to
provide polyfunctionalized diaryl and aryl benzyl ketones.
A. D. Benischke, M. Leroux, I. Knoll, P. Knochel, Org. Lett.,
2016, 18, 3626-3629.

Palladium-catalyzed cross-coupling of aryl boronic acids with acyl chlorides at
room temperature under phosphine-free conditions affords the corresponding aryl
ketones in excellent yields within short times. This synthetic method overcomes
common disadvantages of Friedel-Crafts acylation procedures and is compatible
with a broad range of acyl chlorides.
A. R. Hajipour, R. Pourkaveh,
Synlett, 2014, 25, 1101-1105.

A mild and efficient nickel-catalyzed addition of a broad range of arylboronic
acids to alkyl nitriles in a fluorinated solvent provides various aryl ketones
in very good yields.
D.-H. Tu, Y. Li, B. Zhao, Y.-J. Gu, B. Wang, J.-Y. Lu, J. Lu,
Synlett, 2018, 29, 593-596.

A Ni-catalyzed intermolecular coupling of primary alcohols and
organotriflates provides ketones in the presence of acetone. Two competitive
oxidation processes, that generate aldehyde in situ via hydrogen transfer
oxidation or (pseudo)dehalogenation pathways, are followed by a Ni-catalyzed
carbonyl-Heck process.
T. Verheyen, L. van Turnhout, J. K. Vandavasi, E. S. Isbrandt, W. M. De
Borggraeve, S. G. Newman, J. Am. Chem. Soc.,
2019,
141, 6869-6874.

An array of amides can be cross-coupled with a range of arylboronic acids in
very good yields using a commercially available, air- and moisture-stable
Pd-PEPPSI-IPr complex as a highly reactive precatalyst in a single,
operationally simple protocol. Pd-PEPPSI-IPr provides a significant improvement
over all current Pd/phosphane catalysts for amide N-C bond activation.
P. Lei, G. Meng, Y. Ling, J. An, M. Szostak, J. Org. Chem.,
2017, 82, 6638-6646.

In a protocol for a nickel-catalyzed alkylation of amides, alkylboranes can be
used as nucleophilic partners under mild reaction conditions. The reaction
tolereates various functional groups with respect to both coupling partners.
X. Liu, C.-C. Hsiao, L. Guo, M. Rueping, Org. Lett.,
2018, 20, 2976-2979.

An NHC-based Pd catalyst enables a Suzuki-Miyaura coupling of aryl esters as
electrophiles to provide various ketone-containing products. This contrasts
known reactions of similar esters that provide biaryls via nickel catalysis.
T. B. Halima, W. Zhang, I. Yalaoui, X. Hong, Y.-F. Yang, K. N. Houk, S. G.
Newman, J. Am. Chem. Soc., 2017,
139, 1311-1318.

An efficient palladium-catalyzed addition of arylboronic acids to aliphatic
nitriles provides a broad range of alkyl aryl ketones in good yields. It is
noteworthy that sequential addition and intramolecular annulation reactions of
2-(2-hydroxyphenyl)acetonitriles with arylboronic acids smoothly afford
2-arylbenzofurans in good yields under the standard conditions.
X. Wang, X. Wang, M. Liu, J. Ding, J. Chen, H. Wu, Synthesis, 2013, 45,
2241-2244.

A palladium-catalyzed addition of potassium aryltrifluoroborates to aliphatic
nitriles leads to a wide range of alkyl aryl ketones with good yields. The scope
of the developed approach is successfully explored toward the one-step synthesis
of 2-arylbenzo[b]furans via sequential addition and intramolecular
annulation reactions. The methodology accepted a wide range of substrates and is
applicable to library synthesis.
X. Wang, M. Liu, L. Xu, Q. Wang, J. Chen, J. Ding, H. Wu, J. Org. Chem., 2013,
78, 5273-5281.

A convenient and efficient method allows the synthesis of various arylketones by
nickel-catalyzed addition of arylboronic acids to nitriles. A catalytic cycle
involving Ni(II) species as the catalytic intermediates is proposed.
Y.-C. Wong, K. Parthasarathy, C.-H. Cheng, Org. Lett., 2010,
12, 1736-1739.

A NiCl2(DME)/dppp/Zn system catalyzed an intermolecular insertion of
aryl iodides to nitriles to provide variously substituted arylketone
derivatives in good yields with tolerance of a wide variety of functional groups.
J.-C. Hsieh, Y.-C. Chen, A.-Y. Cheng, H.-C. Tseng, Org. Lett., 2012,
14, 1282-1285.

In a palladium-catalyzed one-pot procedure for the synthesis of aryl ketones,
triazine esters were coupled with aryl boronic acids to provide aryl ketones in
good yields in the presence of 1 mol % Pd(PPh3)2Cl2
for 30 min.
H. Wu, B. Xu, Y. Li, F. Hong, D. Zhu, J. Jian, X. Pu, Z. Zeng, J. Org. Chem.,
2016,
81, 2987-2992.

A highly chemoselective, palladium(0)-catalyzed, direct cross-coupling between
boronic acids and geometrically activated amides shows excellent functional
group tolerance, and delivers the versatile ketone products in high yields.
Notably, the method provides direct access to acyl-metal intermediates from
sterically distorted, bench-stable amide precursors under mild catalytic
conditions.
G. Meng, M. Szostak, Org. Lett.,
2015,
17, 4364-4367.

Nickel catalysis and a site-selective N,N-di-Boc activation enable a
general Negishi acylation of primary amides. The reaction is promoted by a
bench-stable, inexpensive Ni catalyst and shows excellent functional group
compatibility.
S. Shi, M. Szostak, Org. Lett.,
2016, 18, 5872-5875.

The use of CHCl3 as a convenient and safe carbonyl source in the
presence of KOH enables a modular, palladium catalyzed synthesis of
aryl(hetero)aryl benzophenones and aryl benzoates from aryl(hetero)aryl halides
via an initial carbonylation to generate an aroyl halide, which undergoes
coupling with arylboronic acids, boronates, and phenols. Direct carbonylative
coupling of indoles at the third position has also been accomplished.
P. Sharma, S. Rohilla, N. Jain, J. Org. Chem.,
2017, 82, 1105-1113.

Palladacycles catalyze a carbonylative Suzuki-Miyaura coupling of aryl iodides
with arylboronic acids with high turnover numbers and turnover frequencies.
Comparison of the palladacycles with a conventional palladium source shows their
superiority.
P. Gautam, B. M. Bhanage, J. Org. Chem.,
2015,
80, 7810-7815.

Highly selective carbonylative Suzuki reactions of aryl iodides with arylboronic
acids using an in situ generated nanopalladium system under ambient conditions
and in the absence of an added ligand furnished products in high yields. The
addition of pivalic acid can effectively suppress an undesired Suzuki coupling.
The synthesis can be easily scaled up, and the catalytic system can be reused up
to nine times.
Q. Zhou, S. Wei, W. Han, J. Org. Chem., 2014,
79, 1454-1460.

A palladium(II) chloride catalyzed mild cross-coupling of arylboronic acids with
carboxylic anhydrides or acyl chlorides in water in the presence of the
inexpensive surfactant sodium dodecyl sulfate (SDS) gives aryl ketones in good
yields without the use of phosphine ligands.
B. Xin, Y. Zhang, K. Cheng, Synthesis, 2007,
1970-1978.

A direct cross-coupling reaction of aromatic aldehydes with potassium
trifluoro(organo)borates afforded ketones in high yields and under mild
conditions in the presence of a rhodium catalyst and acetone.
M. Pucheault, S. Darses, J-P. Genet, J. Am. Chem. Soc.,
2004,
126, 15356-15357.

A new palladium-catalyzed cross-coupling reaction between arylboronic
acids and mixed anhydrides, generated in situ from carboxylic acids and
pivalic anhydride, is presented.
L. J. Goossen, K. Ghosh, Eur. J. Org. Chem., 2002,
3254-3267.

A new Ni catalyst is capable of effecting the rapid cross-coupling of acid
fluorides, acid chlorides, acyl cyanides, anhydrides, thioesters, and
pyridyl esters with both groups of diorganozinc reagents. Reactions with
acid fluorides as electrophilic partners tolerate epimerizable functionality
as well as leaving groups.
Y. Zhang, T. Rovis, J. Am. Chem. Soc.,
2004,
126, 15964-15965.

A mild and efficient palladium-catalyzed ligand-free decarboxylative coupling of
aryl α-keto carboxylic acid with aryl diazonium fluoroborate provides
unsymmetrical diaryl ketones in good yields via a radical pathway involving an
acyl radical.
S. Panja, P. Maity, B. C. Ranu, J. Org. Chem., 2018, 83,
12609-12618.

Palladium-catalyzed couplings of aryl bromides with N-tert-butylhydrazones
as acyl anion equivalents occurs at the C-position to form N-tert-butyl
azo compounds. Isomerization of these azo compounds to the corresponding
hydrazones, followed by hydrolysis, gave the desired mixed alkyl aryl ketones.
A. Takemiya, J. F. Hartwig, J. Am. Chem. Soc., 2006,
128, 14800-14801.

Pd-catalyzed cross-coupling
reaction of acylindium reagents generated in situ from easily available acid
chlorides and indium with aryl iodides, triflates
and alkenyl triflate enables a reversed-polarity synthesis of various unsymmetric aryl aryl and
alkenyl aryl ketones.
D. Lee, T. Ryu, Y. Park, P. H. Lee, Org. Lett., 2014,
16, 1144-1147.

The use of Zn powder in the presence of LiCl in THF allows a simple,
high-yielding preparation of a broad range of functionalized aryl- and
heteroarylzinc reagents. The synthesis of alkylzinc reagents was performed from
inexpensive alkyl bromides.
A. Krasovski, V. Malakhov, A. Gavryushin, P. Knochel, Angew. Chem. Int. Ed.,
2006,
45, 6040-6044.

Chemoselective cross-coupling of aliphatic and aromatic acyl chlorides with
organostannanes provides a useful alternative to Friedel-Crafts acylations for
the synthesis of ketones. A range of ketones are obtained in high yield using
2.5 mol % of bis(di-tert-butylchlorophosphine)palladium(II) dichloride as
precatalyst. The catalyst tolerates various functional groups including aryl
chlorides and bromides that usually undergo oxidative addition to palladium
complexes.
R. Lerebours, A. Camacho-Soto, C. Wolf, J. Org. Chem., 2005,
70, 8601-8604.

The palladium-catalyzed coupling of 2-pyridyl
esters with organoboron compounds is compatible with various functional groups
and proceeds under mild reaction conditions. The coordination of the nitrogen
atom to Pd is a key step for an efficient reaction.
H. Tatamidani, F. Kakiuchi, N. Chatani, Org. Lett., 2004, 6,
3597-3599.

An addition of arylboronic acids to nitriles catalyzed by a
bipyridine-cyclopalladated ferrocenylimine complex can be conducted in aqueous
media under acidic or basic conditions. The addition of NH4Cl has a
drastic effect on the reaction yields.
A. Yu, J. Li, M. Cui, Y. Wu, Synlett, 2007, 3063-3067.

A mild and general protocol for the carbonylative Suzuki-Miyaura cross-coupling
of sterically hindered ortho-disubstituted aryl iodides with various aryl
boronic acids provides an array of substituted biaryl ketones in good yield. A
carbonylative Negishi coupling that utilizes alkynyl nucleophiles is also
described.
B. M. O'Keefe, N. Simmons, S. F. Martin, Org. Lett.,
2008,
10, 5301-5304.

Tetraorganoindates, which are easily prepared from 1 eq. of InCl3
and 4 eq. of organometallics, could be employed as effective nucleophilic
cross-coupling partners in Pd-catalyzed carbonylative cross-coupling
reactions with a variety of halides. The present method gave unsymmetrical
ketones in good yields.
S. W. Lee, K. Lee, D. Seomoon, S. Kim, H. Kim, H. Kim, E. Shim, M. Lee,
J. Org. Chem., 2004, 69, 4852-4855.

A highly efficient heterogeneous palladium-catalyzed carbonylative
cross-coupling of aryl iodides with triarylbismuths proceeds smoothly at
atmospheric CO pressure and provides various valuable biaryl ketones with high
atom economy, good to excellent yield, and recyclability of the catalyst.
W. Hao, H. Liu, L. Yin, M. Cai, J. Org. Chem.,
2016,
81, 4244-4251.

N-Thiohydroxy succinimide esters (NTSEs) are versatile reagents,
serving as both acyl and acylthio surrogates for the diverse synthesis of
ketones, thioesters, amides, and acyl disulfides by selective cleavage of
similarly reactive C-S and N-S bonds.
Y.-F. Li, Y.-F. Wei, J. Tian, J. Zhang, H.-H. Chang, W.-C. Gao, Org. Lett.,
2022, 24, 5736-5740.

A copper-catalyzed oxidative cleavage reaction of terminal and internal
alkynes using NFSI and TBHP provides aryl ketone products in good yields. NFSI
not only functioned as N-centered radical precursor but also engaged in
the aryl group migration. Mechanistic studies also suggested the important role
of water.
L. Tang, F. Yang, H. Cheng, C. Tan, C. Jin, H. Chen, Y. Huang, S. Zhang, S.
Zhang, W. Song, J. Tan,
Org. Lett., 2020, 22, 8618-8623.

meso-2,4-Bis(diphenylphosphino)pentane (mBDPP) is an effective
regiocontrolling ligand for palladium-catalyzed internal arylation of
electron-rich olefins in DMSO with no need for any halide scavengers.
S. Liu, N. Berry, N. Thomson, A. Pettman, Z. Hyder, J. Mo, J. Xiao, J. Org. Chem., 2006,
71, 7467-7470.

Ammonium salts that can act as hydrogen-bond donors exert a remarkable
acceleration on the rates of the regioselective arylation of electron-rich
olefins by aryl halides in ionic liquids and common solvents.
J. Mo, J. Xiao, Angew. Chem. Int. Ed., 2006,
45, 4152-4157.

Palladium-catalyzed regioselective Heck arylation of various electron-rich
olefins has been accomplished in imidazolium ionic liquids with a wide range
of aryl bromides and iodides. The reaction proceeded with high efficiency
and remarkable regioselectivity. The chemistry provides a simple, effective
method for preparing branched olefins and contributes to the extension of
the Heck reaction to a wider range of substrates.
J. Mo, L. Xu, J. Xiao, J. Am. Chem. Soc.,
2005,
127, 751-760.

A mild and efficient protocol for palladium-catalyzed oxidative Heck reactions
of arylboronic acids with both electron-rich and -deficient olefins works in the
absence of a base, oxygen, or other external oxidants. With a wide variety of
substrates tolerated, the method broadens the scope of palladium-catalyzed
coupling reactions.
J. Ruan, X. Li, O. Saidi, J. Xiao, J. Am. Chem. Soc., 2008,
130, 2424-2425.

A copper-catalyzed aerobic C-C cleavage of single bonds enables a transformation
of epoxides into ketones.
L. Gu, C. Jin, H. Zhang, L. Zhang, J. Org. Chem., 2014,
79, 8453-8456.

A Pd(II)-catalyzed denitrogenative and desulfinative addition of arylsulfonyl
hydrazides to nitriles provides aryl ketones under mild conditions. The reaction
tolerates many common functional groups, and works well in the presence of
electron-donating and electron-withdrawing substituents.
M. Meng, L. Yang, K. Cheng, C. Qi, J. Org. Chem., 2018, 83,
3275-3284.

A selective acylation of readily accessible organomagnesium reagents with
commercially available esters proceeds in short residence times in continuous
flow. Flow conditions prevent premature collapse of the hemiacetal intermediates
despite noncryogenic conditions, thus furnishing ketones in good yields.
B. Heinz, D. Djukanovic, M. A. Ganiek, B. Martin, B. Schenkel, P. Knochel,
Org. Lett., 2020, 22, 493-496.

In a highly regio- and stereoselective hydroarylation, hydroalkenylation, and
hydrobenzylation of ynol ethers, a Pd-catalyzed reductive addition of
organohalides, including aryl, alkenyl, and benzyl halides, in the presence of
2-propanol gives α,β- and β,β-disubstituted olefinic ethers in good yields.
W. Cui, J. Yin, R. Zheng, C. Cheng, Y. Bai, G. Zhu, J. Org. Chem., 2014,
79, 3487-3493.

In a general method for the preparation of α-hydroxyacetophenones,
functionalized arylmagnesium species are transmetalated to the corresponding
arylzinc intermediates, which undergo Cu(I)-catalyzed reaction with
acetoxyacetyl chloride. Acidic hydrolysis of the acetate group releases the
target α-hydroxyacetophenones with minimal production of undesired polymeric
side products.
M. McLaughlin, J. M. Belyk, G. Qian, R. A .Reamer, C.-y. Chen, J. Org. Chem., 2012,
77, 5144-5148.

A palladium catalyzed carbonylation of aryl iodides using near stoichiometric
amounts of carbon monoxide enables the synthesis of monoprotected
1,3-ketoaldehydes. Besides representing platforms for a variety of heterocyclic
structures, these motives serve as viable precursors for the highly relevant
aryl methyl ketones.
T. M. Gøgsig, D. U. Nielsen, A. T. Lindhardt, T. Skrydstrup, Org. Lett., 2012,
14, 2536-2539.

A highly efficient, practical, and ligand-free palladium-catalyzed
carbonylation of aryl iodides with alkenylboronic acids provides a variety of
chalcones and α-branched enones in good yields under an ambient pressure of CO
at room temperature. Moreover, the transformation proceeds well in the presence
of a substoichiometric amount of base.
L. Tang, Y. Gao, J. Chen, L. Yang, B. Xiao, G. Shen, Y. Ouyang, W. Han, Synlett, 2023,
34,
1280-1284.

Silylated cyanohydrins of iodo-substituted aryl, heteroaryl, or cycloalkenyl
ketones undergo an I/Mg-exchange using i-PrMgCl·LiCl. After
subsequent reactions with electrophiles, a facile deprotection
produces polyfunctional ketones in good overall yiels. An extension to
aromatic iodoaldehydes is described.
C.-Y. Liu, H. Ren, P. Knochel, Org. Lett.,
2006,
8, 617-629.

Highly regioselective and efficient Heck reactions of aryl triflates with N-acyl-N-vinylamines
were achieved using Pd2(dba)3, dppf, and diethylisopropylamine in dioxane. The coupling products easily
underwent acidic hydrolysis to the corresponding aryl methyl ketones or in situ
hydrogenation in the presence of (Ph3P)3RhCl under a
hydrogen atmosphere to provide the N-acyl derivatives of benzylic amines.
A. L. Hansen, T. Skydstrup, J. Org. Chem., 2005,
70, 5997-6003.

A new method for the synthesis of organoborates starting from
α,β-unsaturated acetals was developed. The available alkoxy-functionalized
butadienyl- and styrylboronic esters cross couple effectively with a broad
spectrum of aryl halides under mild conditions. The new methods also provide
an effective approach to aromatic ketones.
P. B. Tivola, A. Deagostino, C. Prandi, P. Venturello, Org. Lett.,
2002, 4, 1275-1277.

1-Siloxy-1-alkenylcopper species were generated by 1,2-Csp2-to-O
silyl migration of the copper enolates of acyltriphenylsilanes. The
alkenylcopper species reacted with methyl, benzyl, allylic, tributylstannyl
halides and in the presence of Pd(0) catalyst with aryl and alkenyl iodides
to give geometrically pure (Z)-enol silyl ethers.
A. Tsubouchi, K. Onishi, T. Takeda, J. Am. Chem. Soc., 2006,
128, 14268-14269.

The first enantioselective desymmetrizations of anhydrides with carbon-based
nucleophiles nucleophiles by employing (-)-sparteine as a chiral ligand are
described. These C-C bond-forming reactions proceed in good enantioselectivity
for a range of anhydrides.
R. Shintani, G. C. Fu, Angew. Chem. Int. Ed., 2002, 41,
1057-1059.

The use of titanium(IV) chloride as a Lewis acid for direct ortho-acylation
of phenols and naphthols proves to be a convenient, more general and direct
route to various hydroxyaryl ketones. The route is regioselective, leading
to ortho C-acylated products in satisfactory to high yields in most cases.
A. Bensari, N. T. Zaveri, Synthesis,
2003, 267-271.

tert-Butyl isocyanide insertion enables a simple and efficient
palladium-catalyzed carbonylative Sonogashira coupling. This methodology
demonstrates the utility of isocyanides in intermolecular C-C bond construction
and provides a novel pathway for the synthesis of alkynyl imines which can
undergo simple silica gel catalyzed hydrolysis to afford alkynones. The approach
is tolerant of a wide range of substrates and applicable to library synthesis.
T. Tang, X.-D. Fei, Z.-Y. Ge, Z. Chen, Y.-M. Zhu, S.-J. Ji, J. Org. Chem., 2013,
78, 3170-3175.

A palladium-catalyzed carbonylative Sonogashira coupling of aryl bromides using
near stoichiometric amounts of carbon monoxide converts a broad substrate scope
in good yields. The formed alkynone motive serves as a platform for the
synthesis of various heterocyclic structures, including pyrimidines. Furthermore,
the presented strategy allows effective 13C labeling.
K. T. Neumann, S. R. Laursen, A. T. Lindhardt, B. Bang-Andersen, T. Skrydstrup, Org. Lett., 2014,
16, 2216-2219.

A Pd-catalyzed copper-free carbonylative Sonogashira coupling reaction at
room temperature was achieved by using water as a solvent under balloon
pressure of CO with Et3N as a base.
B. Liang, M. Huang, Z. You, Z. Xiong, K. Lu, R. Fathi, J. Chen, Z. Yang,
J. Org. Chem., 2005,
70, 6097-6100.

Carbonylative coupling of terminal alkynes with aryliodides in the
presence of PdCl2(PPh3)2 as catalyst in 2
eq. of 0.5 M aqueous ammonia, and CO (1 atm) gives the corresponding
α,β-alkynyl ketones in good yields.
M. S. M. Ahmed, A. Mori, Org. Lett., 2003, 5,
3057-3060.

Starting from readily available aryl iodides and allenes, with formic acid as
the CO source and reductant, good yields of α-branched enones were isolated. The
use of a CO source avoids the manipulation of CO gas.
H.-Q. Geng, L.-C. Wang, C.-Y. Hou, X.-F. Wu,
Org. Lett., 2020, 22, 1160-1163.

A practical, convenient, and highly selective Pd-catalyzed addition of
organoboron reagents to dinitriles provides β-ketonitriles with excellent
functional-group tolerance and a broad scope of substrates.
G. Zheng, J. Liu, Y. Shao, F. Zhang, Z. Chen, N. Lv, J. Chen, R. Li, J. Org. Chem., 2021, 86,
861-867.

3-Oxo-3-(hetero)arylpropanenitriles can be synthesized via a carbonylative
palladium-catalyzed α-arylation of tert-butyl 2-cyanoacetates with (hetero)aryl
bromides followed by an acid-mediated decarboxylation step. Through the
combination of only a stoichiometric loading of carbon monoxide and mild basic
reaction conditions for the deprotonation step, an excellent functional group
tolerance was ensured.
M. T. Jensen, M. Juhl, D. U. Nielsen, M. F. Jacobsen, A. T. Lindhardt, T.
Skrydstrup, J. Org. Chem.,
2016,
81, 1358-1366.

Acyl substitution of readily accessible α-siloxy Weinreb amides with
organolithium compounds provides diverse cross-benzoin and α-siloxy ketone
products. Enantiopure benzoins can be accessed via a chiral pool approach.
C. N. Larcombe, L. R. Malins, J. Org. Chem., 2022, 87,
9408-9413.

A concerted metallophotoredox catalysis enables an efficient decarboxylative
functionalization of α,β-unsaturated carboxylic acids with aryl iodides in the
presence of perylene bisimide dye to afford 1,2-diketones.
S. Chand, A. K. Pandey, R. Singh, K. N. Singh, J. Org. Chem., 2021, 86,
6343-6350.

A convenient Pd-catalyzed reaction of aryl halides, tert-butyl isocyanide as the CO
source, and organoaluminum reagents provides 1,2-diketones in very good yields
and with good
functional group tolerance.
B. Chen, X.-F. Wu,
Org. Lett., 2020, 22, 636-641.

Benzil derivatives such as diaryl 1,2-diketones are synthesized via a direct
copper-catalyzed decarboxylative coupling reaction of aryl propiolic acids with
aryl iodides followed by an oxidation. The reaction shows good functional group
tolerance toward ester, aldehyde, cyano, and nitro groups. In addition,
symmetrical diaryl 1,2-diketones are obtained from aryl iodides and propiolic
acid in the presence of palladium and copper catalysts.
H. Min, T. Palani, K. Park, J. Hwang, S. Lee, J. Org. Chem., 2014,
79, 6279-6285.

A general Pd-catalyzed double carbonylation of aryl iodides with secondary or
primary amines provides α-ketoamides in high yields with excellent
chemoselectivities at atmospheric CO pressure. This transformation proceeds
successfully even at room temperature and in the absence of any ligand and
additive.
H. Du, Q. Ruan, M. Qi, W. Han, J. Org. Chem.,
2015,
80, 7816-7823.

A tandem metal-free oxidative aryl migration/C-C bond-cleavage reaction,
mediated by hypervalent iodine reagent, provides straightforward access to
important α-ketoamide and α-ketoester derivatives from readily available acrylic
derivatives via a concerted process of 1,2-aryl shift concomitant with C-C bond
cleavage.
L. Liu, L. Du, D. Zhang-Negrerie, Y. Du, K. Zhao, Org. Lett.,
2014,
16, 5772-5775.

The Pd-catalyzed reaction between aryl or vinyl iodides and ethyl diazoacetate
(EDA) provides cross-coupling products in good yields.
C. Peng, J. Cheng, J. Wang, J. Am. Chem. Soc., 2007,
129, 8708-8709.

A Regel-type transition-metal-free direct C-2 aroylation of (benzo)oxazoles, (benzo)thiazoles
and 1,3,4-oxadiazoles with acid chlorides is catalyzed by N,N-dimethyl-4-aminopyridine
(DMAP) and affords the corresponding 2-ketoazoles in good yields.
P. Lassalas, F. Marsais, C. Hoarau, Synlett, 2013, 24,
2233-2240.



