Tuesday, January 15, 2008
Doris Dallinger
Karl-Franzens-University Graz
Ru-Catalyzed Aryl Azide-Alkyne Cycloaddition
The synthesis of 1,2,3-triazoles via Ru-catalyzed aryl azide-alkyne cycloadditions was described by the group of Valery Fokin from The Scripps Research Institute (Org. Lett. 2007, 9, 5337. DOI: 10.1021/ol701912s). In comparison to the Cu(I)-catalyzed azide-alkyne cycloaddition where 1,4-disubstituted triazoles are obtained, the Ru-catalyzed version produces 1,5-regioisomers of 1,2,3-triazoles. [Cp*RuCl]4 proved to be superior in activity than the original Cp*RuCl(PPh3)2 catalyst and in combination with microwave heating higher yields, cleaner products and shorter reaction times were accomplished. By-product formation and lower yields were noticed under conventional heating conditions in an oil bath.

One-Pot Suzuki-Miyaura/Etherification Reaction
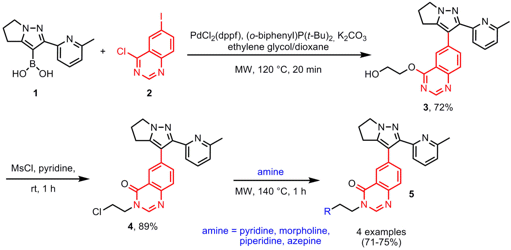
In the course of the synthesis of quinazolinone TGF-β RI inhibitors 5, Hong-yu Li and co-workers from the Lilly Corporate Center in Indianapolis have disclosed the synthesis of key intermediate 3 via a one-pot three-component Suzuki-Miyaura/etherification sequence (Tetrahedron 2007, 63, 11763. DOI: 10.1016/j.tet.2007.08.069). In order to prevent side reactions in the Suzuki coupling of boronic acid 1 and quinazoline 2 (in particular Suzuki reaction at the 2-chloro position of 2), ethylene glycol was employed for the etherification step at the 2-position of 2. In addition, product purification was improved by performing the reaction sequence in one pot. The final products 5 were obtained by amination of scaffold 4, again using microwave heating.
Synthesis of Potential β-Sheet Nucleators via Suzuki Coupling
Vincenzo Santagada and his group from Universitá di Napoli “Federico II”, Italy, have reported on the synthesis of biphenyls 3 that incorporate an amino acid moiety with the carboxyl and amino function in ortho position (Tetrahedron 2007, 63, 12779. DOI: 10.1016/j.tet.2007.09.071). These biphenyl scaffolds were synthesized in order to develop new compounds that are able to induce β-hairpin folding in peptides. Biphenyls 3 were obtained in a single step by the Suzuki coupling of boronic acids 1 and aniline (n = 0), benzylamine (n = 1) or phenethyl amine (n = 2) bromides 2 in aqueous DMF as solvent and 30 min microwave heating at 150 °C.

Asymmetric Heck Reactions using Phosphine-Thiazole Ligands
Pher Andersson and co-workers from the Department of Biochemistry and Organic Chemistry in Uppsala have made investigations toward the impact of the ligand structure on the asymmetric Heck coupling of 2,3-dihydrofuran with various triflates (Adv. Synth. Catal. 2007, 2595. DOI: 10.1002/adsc.200700390). With all employed phosphine-thiazole ligands 1-6 the regioselectivity was excellent whereas the enantioselectivity and the reaction rate were dependent on the substitution patterns of the ligands. For the coupling with aryl triflates bulkier substituents in the 2-position of the ligands enhanced the enantioselectivity and reaction rate: ligand 6 proved to be the best regarding ee´s (94-98%) and reaction time (1 h) whereas with ligand 4 low conversions (28-30%) and ee´s (77-80%) were obtained. By employing microwave heating the reactions proceed faster with constant high enantioselectivities.
.
D. Dallinger, Org. Chem. Highlights 2008, January 15.
URL: https://www.organic-chemistry.org/Highlights/2008/15January.shtm
