Practical Synthesis of Natural Amino Acid Derivatives: Hf(OTf)4-Catalyzed Mannich-Type Reaction of Ketene Silyl Acetals or Enol Silyl Ethers with N,O-Acetals as a Glycine Cation Equivalent
Norio Sakai*, Asuka Sato, Takeo Konakahara
*Department of Pure and Applied Chemistry, Faculty of Science
and Technology, Tokyo University of Science (RIKADAI), Noda, Chiba 278-8510,
Japan, Email: sakachem rs.noda.tus.ac.jp
rs.noda.tus.ac.jp
N. Sakai, A. Sato, T. Konakahara, Synlett, 2009, 1449-1452.
DOI: 10.1055/s-0029-1216743

Abstract
The Hf(OTf)4-catalyzed Mannich-type reaction of an enol silyl ether or a ketene silyl acetal with an N,O-acetal allows the preparation of amino acid derivatives. In particular, use of the N,O-acetal having a bis(trimethylsilyl)amino group directly produced N-unprotected aspartic acid derivatives after a standard aqueous workup.
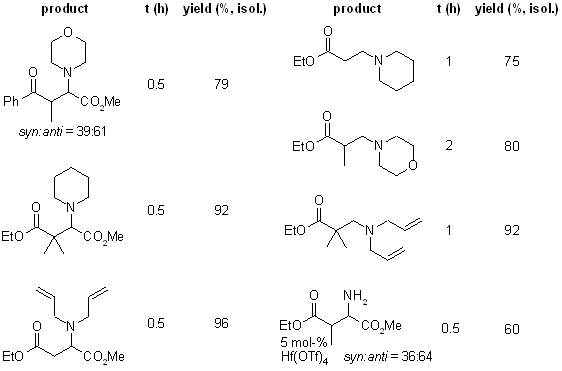
see article for more examples
Key Words
Mannich-type reaction, N,O-acetals, α-amino acids, β-amino acids, hafnium triflate
ID: J60-Y2009-1670
