Totally Synthetic by Paul H. Docherty, 6 February 2008
Total Synthesis of Quinine
Woodward, Williams
R. B. Woodward, W. E. Doering, J. Am. Chem. Soc. 1944, 66, 849-849.
DOI: 10.1021/ja01233a516
R. B. Woodward, W. E. Doering, J. Am. Chem. Soc. 1945, 67, 860-874.
DOI: 10.1021/ja01221a051
A. C. Smith, R. M. Williams, Angew. Chem. Int. Ed. 2008, 47, 1736-1740.

This it’s certainly one of the most interesting chapters of synthetic chemistry, because serious controversy has now been resolved. The whole affair revolves around Woodward’s formal total synthesis of quinine, first published in 1944. His work with Doering led to an advanced intermediate, d-quinotoxine, which had been featured in a total synthesis of quinine performed by Rabe and Kindler in 1918. However, some concern was voiced as the original 1918 paper was somewhat scant on detail. Indeed, there wasn’t a proper experiment - only a reference to the use of similar chemistry in analogous work.
Though this might seem to stitch these works together, one Gilbert Stork was still rather disgruntled with this work. (A full and interesting discussion of the history, politics and science of this affair can be read in Jeffrey Seeman’s awesome paper in Angewandte) Somewhat interestingly, Stork originally voiced his opinion in 1944 by way of an unanswered letter to Woodward asking
“…Would you also tell me whether Rabe’s conversion of quinotoxine into quinine has been repeated by you in your recent work”
With no answer recieved, I guess Storks concern grew, along with his interest in the molecule. His own work on Quinine was complete in 2001 with a publication titled “The First Stereoselective Total Synthesis of Quinine” detailing Storks impressive stereoselective synthesis of the molecule. However, in that paper he once again suggests that the Rabe synthesis is flawed, and that there is no evidence that the chemistry works. With the Rabe work in question, the Woodward synthesis did indeed seem to be in doubt.
Recently, though, vindication was achieved for both Woodward and Rabe, as Williams has recently completed the Rabe chemistry “deploying the experimental protocols originally described by Rabe and his co-workers”. So, we finally get to some chemistry!
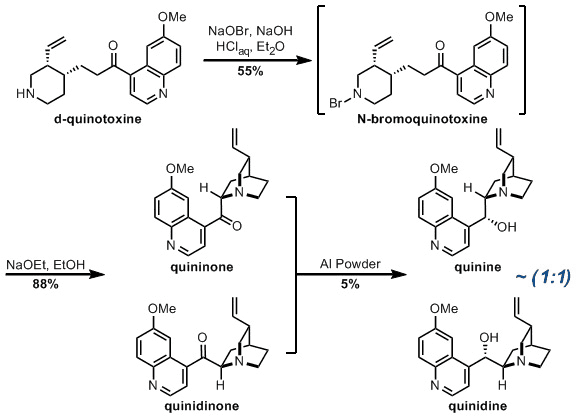
The Rabe chemistry begins with d-quinotoxine, available from an acidic degradation of quinine itself (thus turning this work into a sort of relay synthesis). The starting material was oxidised to the N-bromide, and the bromine displaced with the enolate of the ketone using a base. This resulted in exclusive formation of quinidinon, which was found to isomerise to an equilibrium mixture of the desired quininone and quinidinone. Then to the crux of the problem - the reduction of the ketone to deliver quinine. Rabe states that they used “powdered aluminium” to perform this, but Williams couldn't isolate any product.However, with some aged Al powder - indeed quinine, although in a very modest yield and as a mixture of diastereoisomers.
Woodward needed a few steps making his starting material. Remember: no NMR, flash chromatography or TLC in 1940. An elimination gave an exo-cyclic methylene, which was reduced. Further hydrogenation reduced the pyridine entirely. The amine was protected, and then the remaining aryl group reduced again! Without all the aromaticity, the alcohol was oxidised to form the corresponding cyclohexanone as a mixture of cis/trans-decalins.

Then, a bit of Woodward genius: a nitrite ester ring clevage which is explained in Classics in Total Synthesis II. The oxime was then reduced and eliminated, and after protecting group transformation, a Claisen condensation followed by decarboxylation was used to append the quinoline moiety, completing the formal synthesis. For sure!

Some thanks to Jeffrey Seeman for the paper I mentioned above, and an amazing lecture he gave on this subject last year in Oxford. Also, I want to reaffirm my admiration for Gilbert Stork - good scientists are critical of the literature.
Selected Comments

This paper is indeed remarkable for a several reasons:
1) The authors were actually able to reproduce both the Rabe-Kindler reduction protocol *and* the isolation / crystallization of quinine from the mixture. The art of obtaining pure, crystalline products from very difficult mixtures is, unfortunately, a skill where the chemists of today do not always excel. As stated in the paper: “We must despair that the mastery of crystallization techniques in the early part of the 20th century certainly surpasses that routinely practiced today.”
2) In my past life, I used to work with pyrotechnic mixtures, including various glitter and flitter type formulas. The multitude of sparks, glittering flashes, silvery showers etc. are almost exclusively made with different grades of aluminium. There are spherical powders, stamped flakes, greasy-smelling flakes that leave small silvery specks everywhere, dark pyro grades, and the coarse “Griess” -type powders… each of these behave quite differently, and oftentimes it is very hard to find a good substitute if your favorite brand is no longer supplied. As such, it is almost certain that in 2007 in the USA, one will not find the same brand of aluminium that Rabe and Kindler used in 1918.
Even today, I like to kick off a difficult Grignard with pyro-grade magnesium, fresh from the supplier. What a difference to the old bottle of “Späne nach Grignard zur Synthese”.